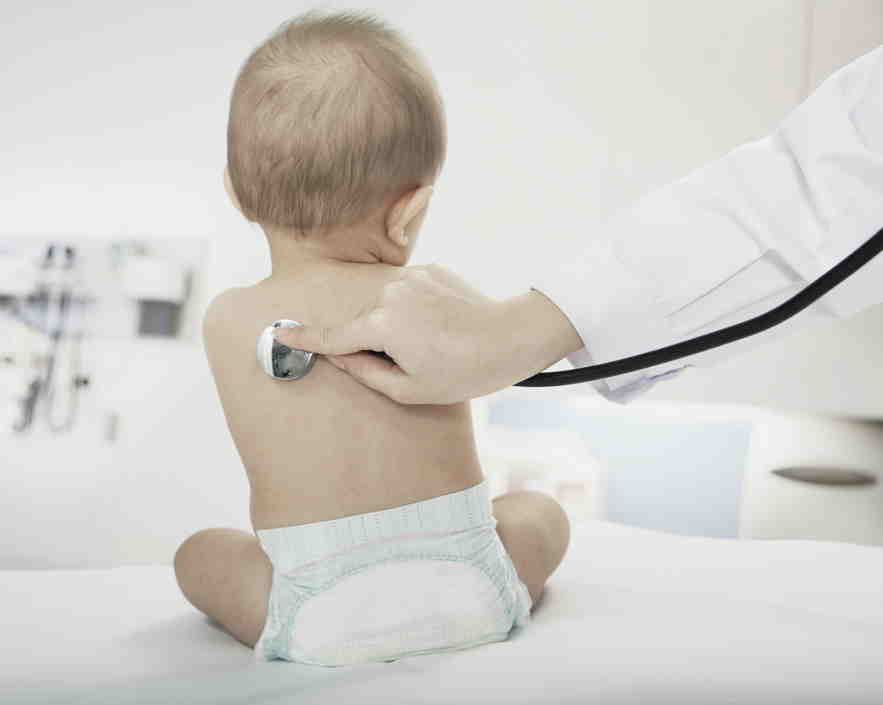
Bubble Baby Families Denied ‘Compassionate Use’ of Live-Saving Therapy
By David Jensen,
Substack
| 05. 25. 2021
Update 5/28: Orchard Exits Life-Saving Bubble-Baby Research in Wake of Flap Over Patient Access
A gene therapy company backed by millions of dollars from the state of California has withheld “compassionate use” of a life-saving therapy sought by parents of children suffering from what is known as the bubble baby disease, according to a patient advocate group and parents.
The families sounded an “SOS” this spring in a letter to the company, Orchard Therapeutics PLC.
“We implore you to treat this as a desperate call for help to save young lives,” the letter said. More than 20 young children are in “urgent need of gene therapy for ADA-SCID (the medical term for the bubble baby disease),” the letter said. (See text here.)
Disclosure of the rejection of the compassionate use treatment is a new twist in the story about the gene therapy, which has saved some 50 lives during its experimental development for bubble babies. The news is likely to intensify criticism of the company for its financially driven decisions concerning the bubble baby disease.
The rare affliction is...
Related Articles
By Priyanka Runwal, Chemical and Engineering News | 08.05.2024
Saritee Sanodiya, 26, has spent countless days wondering if she’ll ever live a “normal” life. Growing up, Sanodiya often missed school, frequenting the hospital for sudden, life-threatening drops in her hemoglobin levels and excruciating pain in her joints. High fever...
It’s been a busy couple of months in biopolitics, with developments in the US, UK, China, Japan, and implicitly on Mars. Time for a brief roundup.
• • •
Bioethics needs an update
The National Research Act is now 50 years old. It was signed into law on July 12, 1974, as a direct response to publicity about the 1932 “Tuskegee Study of Untreated Syphilis in the Negro Male.” The Hastings Bioethics Forum celebrated its anniversary with an...
Image courtesy National Human Genome Research Institute
The U.S. Food and Drug Administration (FDA) is supposed to encourage effective medical advances while also ensuring that patients and research subjects are protected. This dual mandate demands tricky judgment calls that are made more difficult by outside pressures of several kinds, political, judicial, and especially commercial. This April story at Bloomberg examines one deeply troubling pattern of regulatory capture:
Americans Are Paying Billions to Take Drugs That Don’t Work
Companies are increasingly...
By Sarah Kliff and Azeen Ghorayshi, The New York Times | 07.15.2024